ID-FISH Technology is a leading supplier of FISH and ImmunoBlot testing products to clinical Laboratories.
THE ID-FISH ADVANTAGE
For more than 15 years, ID-FISH Technology has been the leader in the development of diagnostic assays using our patented fluorescent in situ hybridization (FISH) and ImmunoBlot technologies. These technologies provide greater sensitivity for the detection of tick-borne diseases.

ID-FISH TECHNOLOGY
ID-FISH powers clinical laboratories through the use of two patented technologies: FISH and ImmunoBlot.
FISH
Fluorescence in situ hybridization (FISH) is a technique used to detect and locate a specific DNA sequence on a chromosome. FISH provides a significant increase in sensitivity and specificity over Giemsa-stained smears.
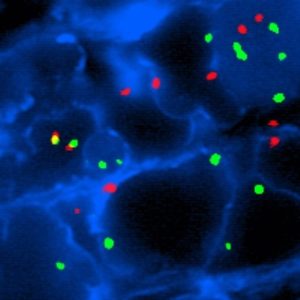
IMMUNOBLOT
ImmunoBlots determine whether pathogen-specific antibodies are present in patient serum or plasma. These tests use recombinant antigens, making them more sensitive and specific than Western Blot or ELISA tests.

Get in Touch
If you would like to learn more about ID-FISH or our products, let us know by completing the form below.

